 Responsible, sustainable, quality product manufacturing. The hardest part of this course of could be successfully communicating together with your factory, as you will be going through language obstacles and time variations. Besides utilizing Blockchain to unravel problems of IoT and Trade four.zero purposes, Tile Knowledge Processing Inc. Manufacturing engineering or manufacturing process are the steps by means of which raw supplies are remodeled right into a final product.
Responsible, sustainable, quality product manufacturing. The hardest part of this course of could be successfully communicating together with your factory, as you will be going through language obstacles and time variations. Besides utilizing Blockchain to unravel problems of IoT and Trade four.zero purposes, Tile Knowledge Processing Inc. Manufacturing engineering or manufacturing process are the steps by means of which raw supplies are remodeled right into a final product.
Working with a producer instantly from the manufacturing facility will open up so many artistic doorways, eradicate undesirable middlemen totally, and ultimately enhance the standard of your product at an reasonably priced value. The title, deal with, and duty of each manufacturer, including contractors, and each proposed production site or facility involved in manufacturing , packaging and testing must be offered.
Hospitality: Our Hospitality enterprise, working as Marietta Hospitality, is a leading North American supplier of visitor facilities supplying a number of national and worldwide resort chains and resorts with single-use private care products resembling shampoos, conditioners, lotions, physique wash and bar cleaning soap.
This is a pre-deliberate scientific method and consists of the initial phases of formulation development, process improvement, setting of process specifications, growing in-process assessments, sampling plans, designing of batch data, defining raw material specs, completion of pilot runs, switch of expertise from scale-up batches to business size batches, listing major course of tools and environmental controls.
Manufacturers who choose to manufacture a sterile product with out terminal sterilization (e.g. aseptic processing) should present sufficient scientific justification and supporting data for the proposed sterilization method. Reference ought to be made to the drug substance data provided in section S.4.4.…
 The Journal of Data Expertise and Laptop Science (JITeCS) is a peer-reviewed open entry journal printed by College of Laptop computer Science, Universitas Brawijaya (UB), Indonesia. Keep away from manufacturing the product if clients aren’t prepared to buy it. Take a look at opponents within the business and understand how your product offers further worth to your prospects. Manufacturing is at all times going to be a continuous process and when business in China relies heavily on relationships, sustaining the connection to your producer can be crucial over time.
The Journal of Data Expertise and Laptop Science (JITeCS) is a peer-reviewed open entry journal printed by College of Laptop computer Science, Universitas Brawijaya (UB), Indonesia. Keep away from manufacturing the product if clients aren’t prepared to buy it. Take a look at opponents within the business and understand how your product offers further worth to your prospects. Manufacturing is at all times going to be a continuous process and when business in China relies heavily on relationships, sustaining the connection to your producer can be crucial over time.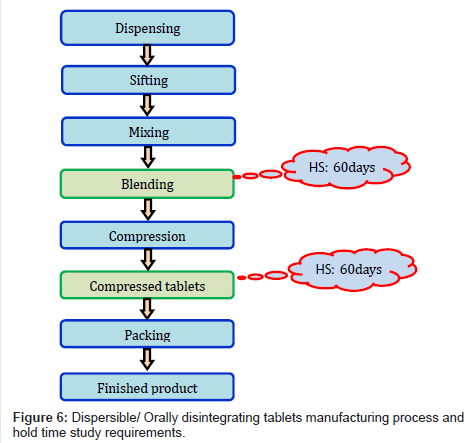 The Journal of Data Know-how and Laptop Science (JITeCS) is a peer-reviewed open entry journal printed by School of Laptop Science, Universitas Brawijaya (UB), Indonesia. The abstract presented in the QOS should include data introduced in a method that it illustrates the soundness conclusions (e.g. solely highest and lowest values recorded in summary, or values that finest represent the info and trends, highest ranges of impurity recorded for all batches at the newest timepoint) and dialogue on the steadiness trends.
The Journal of Data Know-how and Laptop Science (JITeCS) is a peer-reviewed open entry journal printed by School of Laptop Science, Universitas Brawijaya (UB), Indonesia. The abstract presented in the QOS should include data introduced in a method that it illustrates the soundness conclusions (e.g. solely highest and lowest values recorded in summary, or values that finest represent the info and trends, highest ranges of impurity recorded for all batches at the newest timepoint) and dialogue on the steadiness trends. Manufacturing engineering or manufacturing process are the steps through which uncooked supplies are transformed into a ultimate product. Manufacturing is the manufacturing of products to be used or sale using labour and machines , instruments , chemical and biological processing, or formulation, and is the essence of secondary trade The term could seek advice from a range of human activity, from handicraft to high tech , however is most commonly applied to industrial design, by which uncooked supplies from main industry are reworked into completed goods on a large scale.
Manufacturing engineering or manufacturing process are the steps through which uncooked supplies are transformed into a ultimate product. Manufacturing is the manufacturing of products to be used or sale using labour and machines , instruments , chemical and biological processing, or formulation, and is the essence of secondary trade The term could seek advice from a range of human activity, from handicraft to high tech , however is most commonly applied to industrial design, by which uncooked supplies from main industry are reworked into completed goods on a large scale. Sustainable fashion considers the environmental, social and health impression of the design, manufacture and disposal of clothes, footwear and equipment, with the intention to minimise any opposed effects of the enterprise. The difference between having a shopper grab your product from the shelf, or passing it by, stands out as the packaging, and creating nice packaging may be its personal improvement course of. Created in cooperation with Bloomberg Businessweek, the Core77 Design Firm Directory is a great place to begin your seek for an industrial designer and other professionals who may help prepare your product for manufacture.
Sustainable fashion considers the environmental, social and health impression of the design, manufacture and disposal of clothes, footwear and equipment, with the intention to minimise any opposed effects of the enterprise. The difference between having a shopper grab your product from the shelf, or passing it by, stands out as the packaging, and creating nice packaging may be its personal improvement course of. Created in cooperation with Bloomberg Businessweek, the Core77 Design Firm Directory is a great place to begin your seek for an industrial designer and other professionals who may help prepare your product for manufacture. Control strategies are often implemented when growing drugs and manufacturing organic and therapeutic merchandise, nonetheless, increasingly they are thought-about necessary within the manufacture of sterile medicines. For brand new drug submissions (e.g. NDSs, ANDSs, Supplements) concerning drug substances which might be no longer thought of new medicine in line with Part C, Division 8 of the Meals and Drug Laws, consult Well being Canada’s High quality Steerage: Functions for Drug Identification Number Submissions (DINAs) for Prescription drugs for the knowledge that ought to be supplied on the drug substance.
Control strategies are often implemented when growing drugs and manufacturing organic and therapeutic merchandise, nonetheless, increasingly they are thought-about necessary within the manufacture of sterile medicines. For brand new drug submissions (e.g. NDSs, ANDSs, Supplements) concerning drug substances which might be no longer thought of new medicine in line with Part C, Division 8 of the Meals and Drug Laws, consult Well being Canada’s High quality Steerage: Functions for Drug Identification Number Submissions (DINAs) for Prescription drugs for the knowledge that ought to be supplied on the drug substance. SWD Urethane’s OEM focus pushes customized formulations for particular applications to a brand new top. Additionally, this section should determine and describe the formulation and course of attributes (crucial parameters) that may influence batch reproducibility, product performance and drug product high quality. Complementary medical manufacturing services embrace packaging and labeling, quality control, and referrals to certified CMO partners for scientific and industrial scale-up in your preferred country or area.
SWD Urethane’s OEM focus pushes customized formulations for particular applications to a brand new top. Additionally, this section should determine and describe the formulation and course of attributes (crucial parameters) that may influence batch reproducibility, product performance and drug product high quality. Complementary medical manufacturing services embrace packaging and labeling, quality control, and referrals to certified CMO partners for scientific and industrial scale-up in your preferred country or area. Manufacturing engineering or manufacturing course of are the steps by means of which raw supplies are reworked right into a remaining product. Epimed International product manufacturing is well known throughout the medical neighborhood for its dedication to high quality and customer support, and we are able to apply these same rules that can assist you develop and adapt new and present products to fulfill your organization’s needs.
Manufacturing engineering or manufacturing course of are the steps by means of which raw supplies are reworked right into a remaining product. Epimed International product manufacturing is well known throughout the medical neighborhood for its dedication to high quality and customer support, and we are able to apply these same rules that can assist you develop and adapt new and present products to fulfill your organization’s needs. Control methods are often applied when creating medicine and manufacturing biological and therapeutic merchandise, however, more and more they’re thought of important in the manufacture of sterile medicines. The CPID-CE is a condensed version of the QOS and represents the ultimate, agreed upon key knowledge from the drug submission (e.g. checklist of producer(s), manufacturing procedure and management technique, specifications, container closure system together with supply gadgets, storage conditions, retest interval or shelf life, and commitments).
Control methods are often applied when creating medicine and manufacturing biological and therapeutic merchandise, however, more and more they’re thought of important in the manufacture of sterile medicines. The CPID-CE is a condensed version of the QOS and represents the ultimate, agreed upon key knowledge from the drug submission (e.g. checklist of producer(s), manufacturing procedure and management technique, specifications, container closure system together with supply gadgets, storage conditions, retest interval or shelf life, and commitments).